I remember the first time I really understood what the striatum does. Not from a textbook, but from watching a patient with Parkinson’s disease try to initiate movement. She’d sit there, wanting to stand, planning to stand, knowing exactly how to stand—but the command wouldn’t translate into action. Her striatum, depleted of dopamine, couldn’t bridge the gap between intention and execution. That moment crystallized something abstract from neuroanatomy class into something heartbreakingly real: the striatum is where plans become actions, where motivation becomes movement, where wanting something translates into doing something about it.
As a psychologist, I don’t spend my days looking at brain scans or dissecting neural tissue. But understanding the striatum has fundamentally changed how I work with patients struggling with addiction, obsessive-compulsive disorder, ADHD, and movement disorders. Because here’s what’s fascinating about this brain structure: it sits at the intersection of everything that makes us human—our ability to move purposefully, to feel pleasure and motivation, to learn from rewards and punishments, to form habits both helpful and destructive, to make decisions and initiate actions.
The striatum isn’t just one thing doing one job. It’s a complex hub that integrates information from across the brain, processing inputs about what you want, what you’ve learned, what’s rewarding, and what actions are possible, then coordinating outputs that shape your behavior. When it works well, you barely notice it. You decide to pick up your coffee cup, and your hand moves. You feel motivated by a goal, and you take steps toward it. You learn that certain actions lead to good outcomes, and you repeat them. But when the striatum malfunctions—through disease, injury, or dysregulation—the consequences ripple through cognition, emotion, and behavior in ways that profoundly impact quality of life.
What I find most clinically relevant is how the striatum shapes the psychological experiences I encounter daily in my practice. The person with addiction whose reward system has been hijacked? That’s striatal dysfunction. The patient with OCD whose brain gets stuck in repetitive loops? Striatal circuits gone awry. The individual with ADHD who struggles to initiate tasks despite good intentions? Striatal involvement in motivation and action initiation. Understanding the neuroscience doesn’t replace psychological treatment, but it informs it, helping me explain to patients why certain symptoms exist and why particular interventions might help.
Today I want to walk you through what the striatum actually is, how this remarkable structure contributes to everything from walking to wanting, and what happens when things go wrong. This isn’t just academic neuroscience—it’s the foundation for understanding a range of conditions that bring people to therapy, that affect movement and mood, that shape addiction and compulsion. Whether you’re a patient trying to understand your diagnosis, a student learning neuropsychology, or simply curious about how your brain translates desires into actions, understanding the striatum provides crucial insight into what makes behavior possible.
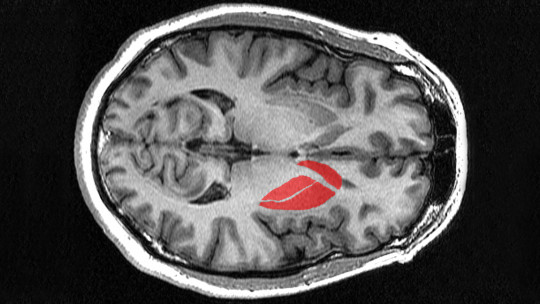
What the Striatum Is: Anatomy and Location
Let’s start with the basics, though even the basics here are more interesting than you might expect. The striatum is the largest structure within the basal ganglia—a collection of interconnected nuclei deep beneath the cerebral cortex. When neuroanatomists say “deep,” they mean it. The striatum sits well below the surface of the brain, nestled in subcortical regions where it can receive information from virtually everywhere in the cortex and send processed outputs to other deep structures that ultimately influence both movement and cognition.
The name “striatum” comes from its striped appearance when you slice it and look at it under basic staining. You see alternating bands of gray matter (neuron cell bodies) and white matter (myelinated axon fibers passing through). This striped pattern isn’t just decorative—it reflects the structural organization that allows the striatum to integrate inputs from across the brain. Those white matter fibers are the internal capsule, carrying information between cortex and subcortical structures, and they physically divide the striatum into distinct regions that have different but related functions.
Anatomically, the striatum divides into two main regions: the dorsal striatum and the ventral striatum. The dorsal striatum, which is what most neuroscientists mean when they say “striatum” without qualification, consists of two structures—the caudate nucleus and the putamen. The caudate nucleus is C-shaped, curving around following the contour of the lateral ventricles. It has a large head at the front, a body that extends back, and a tail that curves down and forward. The putamen sits more laterally, separated from the caudate by that internal capsule I mentioned. Together, the caudate and putamen handle cognitive and motor functions, which we’ll explore shortly.
The ventral striatum occupies the lower portion and includes the nucleus accumbens and the olfactory tubercle. The nucleus accumbens itself divides into a core and shell with slightly different connections and functions. This ventral region is intimately connected with limbic system structures and plays crucial roles in reward processing, motivation, and emotion—functions that have enormous clinical relevance for addiction, depression, and other mood disorders.
Here’s something that matters for understanding clinical conditions: the striatum isn’t uniform. Even within the dorsal or ventral divisions, there are two distinct compartments called the matrix and the striosomes. The matrix makes up most of the striatal volume and receives input from most cortical areas. The striosomes are clusters embedded within the matrix that receive specific inputs from prefrontal cortex and project to different targets. This organization allows the striatum to process information in parallel streams—some handling immediate action selection while others update long-term learning and motivation. When disease affects the striatum, it might impact these compartments differently, producing specific symptom patterns.
The striatum’s location is clinically significant. Being deep in the brain, strokes rarely affect it in isolation—usually if the striatum is damaged, surrounding structures are too. But certain diseases specifically target striatal neurons, producing distinctive syndromes we’ll discuss later. Its position at the hub of multiple brain networks means that striatal dysfunction can produce surprisingly diverse symptoms depending on which connections are disrupted.
Cellular Composition: The Neurons That Make It Work
Now let’s talk about what the striatum is made of at the cellular level, because understanding the neuron types helps explain both normal function and what goes wrong in disease. The striatum isn’t a homogeneous blob—it contains several distinct neuron populations, each with specific roles in striatal function.
The overwhelming majority—somewhere between ninety and ninety-five percent—of striatal neurons are medium spiny neurons, or MSNs. These are the workhorses of the striatum, the projection neurons that send information out to other basal ganglia structures. They’re called “medium” because their cell bodies are intermediate in size, and “spiny” because their dendrites are covered with thousands of tiny protrusions called dendritic spines. These spines are where inputs from cortex and other brain regions make synaptic connections, allowing each MSN to integrate information from up to ten thousand different inputs.
MSNs use GABA as their neurotransmitter, making them inhibitory. This matters enormously for understanding basal ganglia function. The striatum’s output suppresses activity in downstream structures, and the pattern of that suppression—which neurons fire when—shapes what actions get selected and executed. MSNs aren’t just on or off; they exist mostly in a “down state” where they’re relatively quiet, occasionally transitioning to an “up state” where they become active and can fire action potentials. This bistability is crucial for action selection—you need neurons that stay quiet until a clear signal indicates “this is the action to take now.”
Here’s where it gets more interesting: those MSNs aren’t all identical. They divide into two major types based on which dopamine receptor they express and where they project. About half express D1 dopamine receptors and project directly to the output structures of the basal ganglia—the internal segment of the globus pallidus and the substantia nigra pars reticulata. These neurons form what’s called the direct pathway, and when they’re active, they facilitate movement and action. The other half express D2 dopamine receptors and project to the external segment of the globus pallidus, forming the indirect pathway that, through a more complex circuit, suppresses movement and action.
This direct/indirect pathway distinction is fundamental to understanding movement disorders. Parkinson’s disease, for instance, involves dopamine depletion that differentially affects these pathways, reducing direct pathway activity more than indirect pathway activity, leading to difficulty initiating movement. Huntington’s disease involves early selective loss of indirect pathway neurons, producing excessive involuntary movements. The balance between these two pathways determines whether actions get facilitated or suppressed, and disrupting that balance produces the motor symptoms we see in striatal disorders.
The remaining five to ten percent of striatal neurons are interneurons—neurons whose axons stay within the striatum rather than projecting out. Despite being a small minority, these interneurons powerfully influence MSN activity. The largest and most studied are cholinergic interneurons, large neurons that release acetylcholine and help coordinate striatal activity patterns. There are also several types of GABAergic interneurons containing different neurochemicals like parvalbumin or somatostatin. These interneurons help sculpt the timing and pattern of MSN firing, influencing which actions get selected and when.
From a clinical perspective, understanding this cellular composition helps explain why certain drugs work. Dopamine replacement for Parkinson’s targets the dopamine receptors on MSNs. Medications for Huntington’s might target specific receptor types to try rebalancing the pathways. Treatments for addiction might aim to normalize the reward-related plasticity that occurs at synapses onto MSNs. The more we understand the cell types and their specific vulnerabilities, the better we can design interventions.
Functions: What the Striatum Actually Does
This is where neuroscience meets psychology in ways I find most fascinating. The striatum doesn’t do just one thing—it’s involved in multiple functions that span motor control, cognitive processing, and emotional regulation. Let me walk through these functions in ways that connect to experiences you’ll recognize from daily life and clinical conditions.
First and most classically, the striatum is crucial for motor control. Not the basic reflexive movements controlled by spinal cord and brainstem, but voluntary, purposeful movements that you initiate based on goals and plans. The dorsal striatum, particularly the putamen, receives massive input from motor and sensory cortex, processes it in the context of current goals and past learning, and helps select which specific motor programs to execute. When you reach for your coffee cup, the striatum is involved in selecting that action, sequencing the component movements, and adjusting based on sensory feedback about where your hand is relative to the cup.
This motor function becomes starkly obvious when it’s lost. Patients with Parkinson’s disease, where striatal dopamine is depleted, struggle with movement initiation—they might freeze in doorways, unable to start walking despite knowing exactly what they want to do. They have difficulty with movement sequencing—complex multi-step actions fragment into jerky component movements. Their movements become slow and small. This isn’t paralysis—the muscles work fine. It’s a failure of the action selection and execution system that the striatum coordinates.
Beyond basic motor control, the striatum is central to action-outcome learning. You perform an action, something good or bad happens, and the striatum helps encode that association so you’re more likely to repeat rewarding actions and avoid punishing ones. This is operant conditioning at the neural level. Dopamine signals from the substantia nigra and ventral tegmental area report prediction errors—when outcomes are better than expected, dopamine increases; when they’re worse, it decreases. These dopamine signals modify the strength of synapses onto MSNs, gradually shaping which actions get selected in which contexts.
This learning function is where the ventral striatum, particularly the nucleus accumbens, becomes critical. The accumbens is often called the brain’s “reward center,” though that’s oversimplified. It processes reward-related information, motivation, and incentive salience—the quality of “wanting” something that drives goal-directed behavior. When you feel motivated to pursue a goal, when something seems attractive and worth effort, the nucleus accumbens is involved. Addictive drugs hijack this system, producing massive dopamine release in the accumbens that creates powerful “wanting” divorced from actual benefit, driving compulsive drug-seeking even when the person knows it’s harmful.
The striatum is also crucial for habit formation. With repetition, actions that initially required conscious control and striatal selection become automatic habits mediated more by sensorimotor loops through the dorsolateral striatum. This is incredibly efficient—you don’t have to consciously think through every aspect of tying your shoes or driving familiar routes. But it’s also how maladaptive patterns become entrenched. The patient with OCD has striatal loops that keep firing compulsive behaviors despite conscious knowledge they’re unnecessary. The person with addiction has habit circuits that trigger drug-seeking in response to environmental cues even during attempted abstinence.
More recently, research has revealed that the striatum participates in cognitive functions beyond motor control and reward learning. The caudate nucleus in particular receives extensive input from prefrontal cortex and appears involved in executive functions like planning, working memory, and cognitive flexibility. Striatal dysfunction contributes to the executive deficits seen in conditions like ADHD, where action initiation and task switching are impaired. It’s involved in decision-making processes—weighing costs versus benefits, integrating time-delayed outcomes, selecting among competing options.
The striatum also plays a surprising role in social behavior and emotion regulation. The ventral striatum responds to social rewards like positive social feedback, which is why “likes” on social media can be addictive—they trigger the same reward circuits as other reinforcers. Striatal dysfunction is implicated in social deficits seen in autism spectrum disorders and in the motivational deficits characteristic of depression. When the reward system doesn’t fire appropriately, nothing feels worthwhile, and motivation evaporates—that’s anhedonia, a core feature of depression that involves ventral striatal dysfunction.

Connections: The Networks That Make It Work
The striatum doesn’t work in isolation—it’s a hub in multiple brain networks, receiving information from across the cortex and sending processed outputs to other basal ganglia structures. Understanding these connections helps explain how striatal dysfunction produces such diverse symptoms.
The primary input to the striatum comes from cerebral cortex—nearly all cortical areas project to the striatum in an organized way. Motor cortex projects primarily to putamen, cognitive areas of prefrontal cortex project to caudate, limbic cortex projects to ventral striatum. This topographic organization means different striatal regions process different types of information while using similar computational mechanisms. The striatum is essentially doing the same type of processing—action selection through reinforcement learning—but applied to motor actions, cognitive operations, or motivational decisions depending on which cortical areas are providing input.
These cortical inputs use glutamate as neurotransmitter, providing excitatory drive to MSNs. But remember, MSNs are mostly in a down state and require convergent input from many cortical neurons before they’ll fire. This means the striatum is performing a gating function—most potential actions are suppressed most of the time, and only actions with strong convergent support get through the gate.
The other major input to the striatum is dopamine from two midbrain structures: the substantia nigra pars compacta, which projects primarily to dorsal striatum, and the ventral tegmental area, which projects to ventral striatum. This dopamine doesn’t directly excite or inhibit MSNs in simple ways. Instead, it modulates their responsiveness to cortical inputs and shapes synaptic plasticity. Dopamine essentially acts as a teaching signal, indicating when outcomes are better or worse than expected, allowing the striatum to learn from experience.
The timing of dopamine release carries crucial information. When something unexpectedly good happens, dopamine neurons burst fire, flooding the striatum with dopamine that strengthens recently active synapses—”that action was good, do it more.” When expected rewards don’t materialize, dopamine neurons pause their baseline firing, creating a dip in dopamine that weakens those synapses—”that didn’t work as expected, try something else.” This dopamine-mediated learning is how the striatum gradually tunes action selection based on outcomes, slowly shifting behavior toward more rewarding patterns.
The striatum’s outputs go primarily to the globus pallidus and substantia nigra pars reticulata. Remember those direct and indirect pathways? The direct pathway MSNs (D1-expressing) project to GPi and SNr, inhibiting neurons there. Since GPi and SNr normally inhibit thalamus, striatal direct pathway activation releases the thalamus from inhibition, allowing it to activate cortex, facilitating movement and action. It’s a double-negative that produces a positive—inhibiting the inhibitor permits action.
The indirect pathway MSNs (D2-expressing) project to GPe, which through additional synapses in the circuit, eventually increases inhibition from GPi/SNr onto thalamus, suppressing action. So direct pathway activation facilitates “go” while indirect pathway activation promotes “no-go.” The balance between these pathways determines which of many possible actions gets selected for execution.
There’s also a third pathway worth mentioning: the hyperdirect pathway that bypasses the striatum, running from cortex directly to the subthalamic nucleus, which then excites GPi/SNr. This pathway provides rapid global suppression that can pause action selection when something unexpected happens, giving the striatum time to reassess the situation. It’s like a brake that holds everything while your brain figures out what to do next.
Disorders Affecting the Striatum
Now let’s discuss what happens when the striatum doesn’t work properly. Multiple disorders involve striatal dysfunction, producing a range of motor, cognitive, and psychiatric symptoms. Understanding these conditions isn’t just academic—it’s directly relevant to clinical practice and to patients trying to understand their diagnoses.
Parkinson’s disease is the most well-known striatal disorder. It involves progressive degeneration of dopamine neurons in the substantia nigra, depleting dopamine in the dorsal striatum. This creates an imbalance between direct and indirect pathways—with less dopamine, D1 pathway neurons (facilitation) become less responsive while D2 pathway neurons (suppression) become relatively more active. The result is profound difficulty initiating movement, slow movement, rigidity, and resting tremor. Patients describe feeling trapped in their bodies—they know what they want to do, they can plan it mentally, but the motor system won’t respond appropriately.
I’ve worked with several Parkinson’s patients on the psychological aspects of the disease, and the frustration and depression are enormous. The motor symptoms are obvious, but there are also cognitive changes—difficulty with multitasking, planning, and cognitive flexibility—that reflect the role of dopamine and striatal function in executive processes. Depression is extremely common, partly reactive to disability but also reflecting the dopamine depletion affecting reward and motivation circuits. Treatment with levodopa (dopamine precursor) helps motor symptoms but can produce impulse control problems as ventral striatal reward circuits get overstimulated.
Huntington’s disease is a genetic disorder involving progressive degeneration of striatal neurons, particularly MSNs in the indirect pathway initially. Unlike Parkinson’s, where movement is too suppressed, Huntington’s produces excessive, involuntary movements called chorea. The striatum can’t properly suppress unwanted movements, so random movements break through. As the disease progresses and more striatal neurons die, cognitive decline, personality changes, and psychiatric symptoms emerge, reflecting the striatum’s roles beyond just motor control.
What’s particularly tragic about Huntington’s is that it’s a genetic disorder—if you have the gene mutation, you will develop the disease, typically in middle age. Patients often know decades before symptoms start that they carry the gene, creating tremendous psychological burden. There’s currently no cure, only symptomatic treatment. Research is exploring whether interventions targeting the mutant protein might slow progression, but we’re not there yet.
Tourette syndrome involves both motor and vocal tics—sudden, repetitive movements or vocalizations that the person feels compelled to perform. The neurobiology is complex, but abnormalities in striatal circuits, particularly involving the connections between striatum and cortex, appear central. The striatum isn’t properly gating actions, allowing unwanted motor programs to break through. Interestingly, people with Tourette’s describe a premonitory urge before tics—an uncomfortable sensation relieved by performing the tic. This suggests the striatum is generating inappropriate “go” signals that create that urge. Behavioral therapy for Tourette’s teaches competing responses that can suppress tics, essentially training alternative striatal patterns.
Obsessive-compulsive disorder involves striatal dysfunction as a core feature. Brain imaging studies consistently show abnormalities in cortico-striatal-thalamic circuits. The repetitive, compulsive behaviors of OCD resemble habits gone haywire—striatal loops that keep firing the same behavioral sequences despite conscious knowledge they’re unnecessary. The person with OCD knows their fear is irrational, knows washing their hands 50 times isn’t necessary, but the urge is overwhelming because striatal circuits are generating powerful “this action is necessary” signals.
I treat OCD patients with exposure and response prevention, which works by essentially retraining these striatal circuits. We expose patients to anxiety triggers without allowing the compulsion, teaching the striatum that the feared consequence doesn’t occur, that the compulsion isn’t necessary. It’s difficult work—you’re fighting against powerful striatal drive—but it works because you’re leveraging the same learning mechanisms that created the problem to retrain the circuit.
Addiction fundamentally involves hijacking of striatal reward circuits. All drugs of abuse increase dopamine in the nucleus accumbens, far more than natural rewards. This creates powerful reinforcement that drives continued use. Over time, synapses in the accumbens undergo maladaptive plasticity, becoming hypersensitive to drug-related cues while becoming less responsive to natural rewards. The striatum’s learning mechanisms, designed to help organisms pursue beneficial outcomes, instead lock onto drug-seeking as the primary goal. This is why addiction isn’t simply weak willpower—it reflects pathological learning in striatal circuits that’s extremely difficult to override.
Treatment for addiction must address these neurobiological changes. Cognitive-behavioral approaches help patients identify triggers, develop coping strategies, and engage in non-drug rewards to gradually retrain striatal circuits. Medications can help—some reduce drug cravings by normalizing dopamine, others block drug effects to prevent reinforcement. But recovery requires persistent retraining of those deeply ingrained striatal habits, which is why addiction is so challenging and relapse so common.
ADHD involves striatal dysfunction as well, particularly affecting dopamine signaling and striatal-frontal connections. The core symptoms—inattention, impulsivity, hyperactivity—reflect problems with action selection, inhibition, and sustained goal-directed behavior that require intact striatal function. Brain imaging shows reduced striatal volume and altered activation patterns in ADHD. Stimulant medications that increase dopamine help by normalizing striatal signaling, improving ability to focus attention and inhibit inappropriate responses.
Depression includes striatal involvement, particularly in the anhedonia and motivational deficits that characterize the disorder. When nothing feels pleasurable or worthwhile, when you can’t generate motivation to pursue goals, that reflects ventral striatal dysfunction. The reward system isn’t responding normally, so nothing triggers the “wanting” that drives behavior. This is why behavioral activation—scheduling and gradually re-engaging in potentially rewarding activities—helps depression. You’re forcing striatal re-exposure to rewards, allowing the system to relearn that activities can be worthwhile.
Clinical Implications and Treatment Approaches
Understanding striatal function isn’t just interesting neuroscience—it has direct clinical implications for how we assess and treat disorders. Let me share how this knowledge informs my practice and what it might mean for patients dealing with striatal dysfunction.
First, recognizing that many seemingly psychiatric conditions have neurobiological substrates helps reduce stigma and shame. When patients understand that their OCD compulsions reflect abnormal striatal circuit activity rather than moral weakness or character flaw, it’s liberating. When someone with addiction understands their brain’s reward system has been pathologically altered by drug exposure, they can conceptualize treatment as neurobiological retraining rather than simply exerting more willpower. This doesn’t absolve responsibility, but it contextualizes the difficulty and validates why overcoming these conditions requires sustained effort and often professional help.
Assessment benefits from understanding striatal involvement. When I’m evaluating someone with motor symptoms plus cognitive or psychiatric changes, I’m thinking about basal ganglia disorders. When someone describes difficulty initiating actions despite intact knowledge and planning, I’m considering frontal-striatal dysfunction. When habit-like behaviors persist despite attempts to stop, I’m conceptualizing it as potentially involving striatal habit circuits. This neurobiological understanding guides what questions I ask and what recommendations I make.
Treatment planning incorporates striatal neuroscience. For conditions involving striatal dysfunction, I know that interventions targeting those circuits are most likely to help. Exposure-based treatments for OCD and anxiety work because they retrain striatal learning. Behavioral activation for depression works by re-engaging reward circuits. Habit reversal training for tics leverages the striatum’s ability to learn competing responses. Understanding the mechanism makes me more confident in recommending these approaches and helps me explain to patients why they work.
Medication decisions benefit from this knowledge too. When considering medication for ADHD, depression, OCD, or Parkinson’s, understanding how different drugs affect dopamine, serotonin, or other neurotransmitters in striatal circuits helps predict efficacy and side effects. I can explain to patients why stimulants help ADHD—they increase dopamine in striatal circuits involved in attention and action control. I can discuss why SSRIs help OCD—partly through effects on serotonin-dopamine interactions in cortico-striatal circuits. This informed discussion improves treatment adherence.
Importantly, understanding striatal function reveals why some interventions work synergistically. For depression with motivational deficits, combining medication that normalizes neurotransmitter function with behavioral activation that provides structured reward exposure addresses the problem from both neurochemical and learning perspectives. For addiction, combining medications that reduce craving with CBT that retrains behavioral patterns targets both the altered neurochemistry and the learned associations. Comprehensive treatment that addresses multiple aspects of striatal dysfunction is typically more effective than any single intervention alone.
I also use this knowledge to set realistic expectations. Striatal learning mechanisms involve gradual synaptic changes through repeated experience. This means behavioral treatments targeting these circuits require time and repetition. Exposure therapy for OCD doesn’t work after one session—you need repeated exposures to retrain the circuits. Recovery from addiction requires sustained abstinence while the brain slowly readjusts. Understanding the neurobiological timescale helps patients persist through treatment rather than giving up when immediate change doesn’t occur.
FAQs About the Striatum
Where exactly is the striatum located in the brain?
The striatum is a subcortical structure located deep within the brain, beneath the cerebral cortex. It sits in the forebrain on both the left and right sides, forming the largest component of the basal ganglia. Specifically, the caudate nucleus curves along the lateral ventricles, while the putamen sits more laterally, separated from the caudate by white matter tracts of the internal capsule. The nucleus accumbens, part of the ventral striatum, is located at the junction where the caudate and putamen meet at the front of the brain. Because it’s so deep in the brain, the striatum can’t be seen from the surface and is only visible in brain scans or dissected tissue. Its position at the center of multiple brain networks allows it to integrate information from cortical areas throughout the brain.
What is the main function of the striatum?
The striatum’s primary function is action selection and reinforcement learning. It receives information from across the cortex about current goals, context, and sensory input, then helps select which actions to perform based on past learning about what led to rewarding or punishing outcomes. For motor actions, the striatum helps initiate and sequence voluntary movements; for cognitive operations, it supports decision-making and executive functions; for motivational processes, it evaluates rewards and drives goal-directed behavior. Essentially, the striatum bridges the gap between planning and doing, between wanting and acting. It does this through two pathways—a direct pathway that facilitates actions and an indirect pathway that suppresses them—with the balance between these pathways determining what gets executed. Learning occurs through dopamine signals that strengthen or weaken these pathways based on whether outcomes are better or worse than expected.
What happens when the striatum is damaged?
Striatal damage produces different symptoms depending on which region is affected and the extent of damage. Damage to the dorsal striatum, particularly the putamen, typically causes motor problems—difficulty initiating movement, slow movements, poor movement coordination, or sometimes involuntary movements. Damage to the caudate often produces cognitive and executive function deficits—problems with planning, organizing, mental flexibility, and working memory. Ventral striatum damage, particularly to the nucleus accumbens, can cause motivational deficits, anhedonia, difficulty learning from rewards and punishments, and problems with goal-directed behavior. Because the striatum is involved in habit formation, damage can disrupt well-learned automatic behaviors. The specific symptoms depend heavily on which part of the striatum is affected—focal strokes might cause relatively circumscribed deficits, while degenerative diseases affecting the whole structure produce more pervasive problems across motor, cognitive, and motivational domains.
How does dopamine affect the striatum?
Dopamine plays a crucial modulatory role in striatal function. Dopamine neurons from the substantia nigra and ventral tegmental area project to the striatum, where dopamine acts on two main receptor types on medium spiny neurons. D1 receptors on direct pathway neurons are activated by dopamine and facilitate movement and action selection. D2 receptors on indirect pathway neurons are also activated but through a more complex mechanism that involves suppression of competing actions. Critically, dopamine signals when outcomes are better or worse than expected, acting as a teaching signal that strengthens or weakens synapses to guide learning. When rewards exceed expectations, dopamine bursts strengthen recently active connections, making those actions more likely in the future. When rewards fall short, dopamine dips weaken connections. This is how the striatum learns through experience which actions lead to good outcomes. Diseases affecting dopamine, like Parkinson’s, profoundly disrupt striatal function and produce severe motor and cognitive symptoms.
What’s the difference between the ventral and dorsal striatum?
The ventral and dorsal striatum are anatomically distinct regions with different connections and functions, though they use similar computational mechanisms. The dorsal striatum consists of the caudate nucleus and putamen and is primarily involved in motor control and cognitive functions. It receives input from motor, sensory, and association cortex and helps coordinate voluntary movement and executive functions. The ventral striatum consists of the nucleus accumbens and olfactory tubercle and is more closely connected with limbic structures. It processes reward, motivation, and emotion, playing crucial roles in reinforcement learning, goal-directed behavior, and the subjective experience of pleasure and wanting. Functionally, the dorsal striatum asks “how do I do this?” while the ventral striatum asks “is this worth doing?” Together, they coordinate behavior by combining motivational drive from the ventral striatum with motor and cognitive implementation from the dorsal striatum. Many psychiatric conditions involve ventral striatal dysfunction while movement disorders more often affect dorsal striatum, though there’s considerable overlap.
Can the striatum recover from damage?
Recovery from striatal damage depends on the cause and extent of injury. The brain has some capacity for neuroplasticity—surviving neurons can sometimes take over functions of damaged ones, and remaining circuits can reorganize to partially compensate. After stroke affecting the striatum, patients often show some recovery over months through rehabilitation that helps remaining circuits develop alternative strategies. However, in degenerative diseases like Parkinson’s or Huntington’s where neurons progressively die, recovery isn’t possible though treatments can manage symptoms and potentially slow progression. In conditions involving striatal dysfunction rather than structural damage—like OCD or addiction—targeted therapies can retrain abnormal circuits, essentially creating functional recovery by normalizing activity patterns. The striatum’s learning capacity, which creates problems in these disorders, also provides the mechanism for improvement through repeated practice of healthier patterns. Youth generally predicts better recovery due to greater plasticity, while extensive damage in older adults may show limited improvement. Rehabilitation outcomes are best when started early and sustained intensively.
How is the striatum involved in addiction?
The striatum, particularly the nucleus accumbens, is central to addiction. All addictive drugs increase dopamine in the accumbens far beyond levels produced by natural rewards, creating intense reinforcement that powerfully motivates repeated drug use. Over time, this excessive dopamine causes maladaptive changes in striatal neurons—synapses become hypersensitive to drug-related cues while becoming less responsive to natural rewards like food, social connection, or other pleasures. The striatum essentially learns that drugs are the most important goal, overriding normal decision-making and driving compulsive drug-seeking even when the person knows it’s harmful. Habit circuits in the dorsal striatum also become involved, making drug-seeking automatic and difficult to consciously control. Environmental cues associated with drug use trigger intense craving because the striatum has learned those associations. This neurobiological hijacking explains why addiction is so powerful and difficult to overcome—you’re fighting against the brain’s fundamental learning and motivation systems. Treatment must address these striatal changes through both medications that normalize dopamine function and behavioral interventions that retrain striatal circuits toward healthier goals.
Is the striatum the same as the basal ganglia?
No, the striatum is part of the basal ganglia but not identical to it. The basal ganglia are a collection of interconnected subcortical nuclei that include the striatum, globus pallidus, subthalamic nucleus, and substantia nigra. The striatum is the largest component and the main input structure of the basal ganglia, receiving information from cortex and sending processed outputs to other basal ganglia nuclei. Think of the basal ganglia as a whole system for action selection and motor control, with the striatum being the entry point where cortical information enters this system. The globus pallidus and substantia nigra are output structures that send information to thalamus, which then projects back to cortex, completing the loop. The subthalamic nucleus provides an additional pathway. Together, these structures form integrated circuits that coordinate movement, learning, and decision-making. When people talk about “striatal disorders,” they often mean conditions primarily affecting the striatum but recognize that the whole basal ganglia system is involved.
What treatments target the striatum?
Multiple treatments target striatal function directly or indirectly. For Parkinson’s disease, levodopa replaces lost dopamine to improve striatal signaling, while deep brain stimulation of the subthalamic nucleus modulates basal ganglia circuits including the striatum. For addiction, medications like naltrexone block opioid receptors in the striatum to reduce craving, while behavioral therapies retrain reward circuits through exposure to non-drug rewards. For OCD, exposure and response prevention therapy retrains striatal habit circuits by breaking the link between obsessions and compulsions. SSRIs help OCD partly through effects on cortico-striatal circuits. For ADHD, stimulant medications increase dopamine in the striatum, improving attention and impulse control. For depression, behavioral activation re-engages ventral striatal reward circuits. Physical and occupational therapy after stroke help surviving striatal circuits reorganize and compensate. Increasingly, researchers are exploring neurofeedback and brain stimulation techniques to directly modify striatal activity patterns. The specific treatment depends on the disorder and the nature of striatal dysfunction, but understanding striatal involvement guides which interventions are most likely to help.
How does aging affect the striatum?
Normal aging produces gradual changes in striatal structure and function. The striatum shows modest volume loss with age, and dopamine neurons in the substantia nigra and ventral tegmental area decline, reducing dopamine availability. Dopamine receptors on striatal neurons also decrease. These changes contribute to age-related slowing of movements, reduced learning rates for new motor skills, and some decline in reward sensitivity and motivation. However, normal aging doesn’t produce the severe symptoms seen in Parkinson’s or other striatal disorders—healthy older adults show functional decline but maintain adequate striatal function for daily activities. The age-related changes do make older adults more vulnerable to movement disorders and may contribute to late-life depression through effects on reward circuits. Staying physically and mentally active appears protective, potentially through maintaining dopamine function and encouraging neuroplasticity in striatal circuits. When symptoms like severe movement slowing, rigidity, or profound motivational deficits appear, that suggests disease rather than normal aging and warrants medical evaluation. Understanding normal versus pathological striatal aging helps clinicians and patients distinguish expected age-related changes from concerning symptoms requiring intervention.
By citing this article, you acknowledge the original source and allow readers to access the full content.
PsychologyFor. (2025). Striatum: Structure, Functions and Associated Disorders. https://psychologyfor.com/striatum-structure-functions-and-associated-disorders/


